1. Fascia as a functional mechanical environment
The pelvic fascial system constitutes a tissue continuum organizing the mechanical relationship between neural, muscular, and osseous structures. It is not a simple enveloping tissue, but an active mechanical environment whose properties determine how constraints are transmitted, absorbed, or dissipated during movement and postural variations (Stecco & Stecco, 2024).
From a clinical–functional perspective, fascia quality is not assessed by its morphological appearance, but by its capacity to allow fine relative mobility between the structures it connects. This relative mobility is an essential condition for the mechanical tolerance of the pudendal nerve during pelvic, hip, and sitting-related movements.
The nerve–fascia interface, particularly the relationship between the epineurium of the pudendal nerve and the obturator internus fascia, thus represents a major functional determinant. Any alteration of this interface modifies the mechanical regime to which the nerve is exposed, independently of the presence of overt compression or visible lesions.
2. Neurodynamics: gliding as a clinical imperative
Neural gliding refers to the capacity of a nerve to move relative to surrounding tissues during movement. Clinically, this phenomenon represents a fundamental mechanical requirement: the nervous system forms a continuous structure that must constantly adapt to variations in length and trajectory induced by movement (Butler, 2000).
When gliding is preserved, mechanical constraints are diffusely redistributed and remain compatible with neural physiology. The nerve then benefits from a true mobility reserve, allowing it to tolerate significant postural changes without triggering pain signals.
Conversely, loss of relative mobility transforms ordinary actions—sitting down, standing up, walking—into sources of repeated mechanical aggression. The nerve becomes mechanically vulnerable: each movement induces a local increase in tensile and shear forces, progressively lowering the threshold of neuropathic tolerance.
3. Fascial densification and loss of mechanical tolerance
In pudendal neuralgia, a strictly localized analysis of pain rapidly reaches its limits. The classic opposition between so-called “local” pain and “radiating” pain fails to account for clinical presentations in which symptoms fluctuate according to posture, duration of sitting, and repetition of constraints, without identifiable lesion continuity (Labat et al., 2008; Robert et al., 1998).
A functional reading based on tissue continuity makes it possible to move beyond this artificial opposition. Within the pelvic fascial network, mechanical constraints are not transmitted in a punctual manner, but according to a distributed logic: a local alteration in tissue dynamics may modify the global mechanical regime applied to adjacent structures, without direct neural projection or isolated anatomical compression (Stecco & Stecco, 2024).
Fascial densification corresponds to this type of functional alteration. It refers to a progressive modification of the mechanical properties of fascia, characterized by reduced gliding capacity between tissue layers. It is not a lesion, but an adaptation to a mechanically impoverished environment, as observed in situations of sustained postural constraints, particularly prolonged sitting (Stecco & Stecco, 2024).
At the clinical–functional level, fascia can be viewed as a hydrophilic material whose mechanical properties depend on its ability to reorganize under constraint. When this capacity is preserved, tissue layers maintain low relative friction and allow the micro-deformations necessary for adaptation to movement. When this dynamic is reduced, tissue loses its functional fluidity and behaves as a medium with limited reversible deformation, increasing shear forces within tissue interfaces (Stecco & Stecco, 2024).
© Blue Portance — 2026
This transformation of the fascial mechanical regime does not remain confined to the tissue itself. By reducing relative mobility between structures, it alters the mechanical environment of elements that depend on gliding to tolerate movement constraints. The fascial system, organized as a continuous network, then transmits these constraints in a distributed manner, potentially affecting anatomically distinct zones without direct peripheral nerve injury (Stecco & Stecco, 2024).
The pudendal nerve, intimately coupled to these fascial interfaces along its pelvic course, is particularly exposed to such changes. When densification sets in, the nerve is no longer free to perform micro-movements. It is not statically compressed, but subjected to repeated micro-aggressions with each postural variation, especially during transitions to sitting or prolonged sitting (Cholewa et al., 2024).
This situation is sufficient to explain the emergence and persistence of neuropathic pain in the absence of any objectifiable anatomical lesion. Pain does not result from an isolated “pinch,” but from a loss of mechanical compatibility between the nerve and its tissue environment, progressively lowering tolerance to ordinary constraints and favoring symptom chronicization (Cholewa et al., 2024; Stecco & Stecco, 2024).
4. Alcock’s canal: dynamic guidance and functional vulnerability
Alcock’s canal occupies a central place in understanding pudendal neuralgia, precisely because it concentrates multiple factors of mechanical vulnerability without constituting, in itself, a systematically pathological compression zone (Robert et al., 1998; Labat et al., 2008).
From a functional standpoint, Alcock’s canal corresponds to a duplication of the obturator internus fascia, forming a guidance device intended to accompany the pudendal nerve in a region subject to significant mechanical variations (Robert et al., 1998). It is not a passive space, but a dynamic interface designed to allow the nerve to follow pelvic and hip movements while limiting excessive constraints.
This guidance function relies on an essential condition: the nerve’s gliding capacity within its fascial environment. As long as this relative mobility is preserved, the canal plays a protective role, allowing the nerve to adapt to postural changes and pelvic movements without accumulation of local mechanical stress (Butler, 2000).
When fascial dynamics are altered—by densification, adjacent muscle hypertonia, or repeated postural constraints—the functioning of the canal changes profoundly (Stecco & Stecco, 2024; Cholewa et al., 2024). The guidance device loses its adaptive capacity and becomes a zone of increased mechanical friction. The nerve is not statically compressed, but exposed to repeated micro-aggressions with each postural variation.
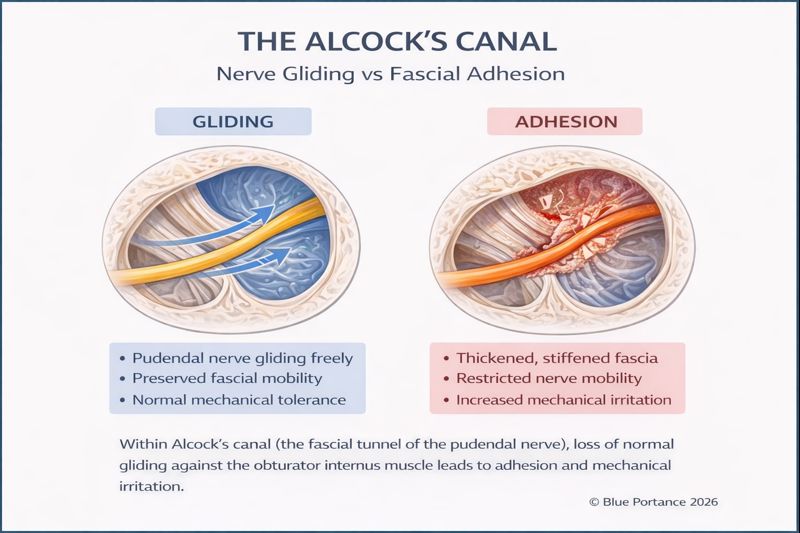
This functional reading explains why Alcock’s canal is frequently involved in symptomatology without any frank stenosis being objectified on imaging (Labat et al., 2008). Pain does not result from an isolated structural conflict, but from defective mechanical adaptation in a zone where the nerve is particularly dependent on tissue interface quality.
Clinically, this vulnerability manifests as pain strongly dependent on posture and duration of exposure. Prolonged sitting, by reducing micro-variations of movement and increasing local constraints, accentuates friction and irritation phenomena within the canal (Cholewa et al., 2024). Conversely, postural variation or unloading phases may transiently improve symptoms, without any anatomical modification of the canal.
Alcock’s canal thus appears not as a unique and autonomous cause of pudendal neuralgia, but as a functional revealer of the state of pelvic tissue dynamics. It represents a passage point where the effects of fascial densification, muscular hypertonia, and loss of neural gliding combine and become clinically perceptible (Stecco & Stecco, 2024).
5. Chronicization of pudendal neuralgia: functional locking and self-sustaining dynamics
Chronicization of pudendal neuralgia cannot be understood through a linear or strictly lesion-based reading. In many cases, persistence of pain is explained neither by anatomical worsening nor by the appearance of a new structural injury, but by the progressive establishment of a mechanical regime unfavorable to the pelvic system (Labat et al., 2008; Robert et al., 1998).
This regime is characterized by a combination of prolonged postural constraints—particularly sitting—and a reduction of the micro-variations necessary to maintain tissue tolerance. Insufficient variation transforms initially reversible constraints into fixed constraints, durably modifying the dynamics of the tissues involved (Stecco & Stecco, 2024).
Functionally, this situation activates several self-sustaining feedback loops. Neuropathic pain induces protective muscular hypertonia, particularly in the deep pelvic muscles. This hypertonia increases local mechanical constraints, reduces relative mobility of fascial interfaces, and aggravates loss of inter-tissue gliding, further exposing the pudendal nerve to repeated micro-aggressions (Cholewa et al., 2024).
In parallel, reduced micro-movements and persistent constraints favor progressive tissue densification. This adaptive response, initially reversible, may evolve toward durable functional stiffening—sometimes described as adaptive fibrosis—further limiting tissue capacity to absorb and redistribute mechanical constraints (Stecco & Stecco, 2024).
These mechanisms are not independent phenomena, but part of a systemic organization. Loss of mechanical tolerance of the pudendal nerve is not the consequence of a single event, but the result of a lasting imbalance between postural constraints, tissue adaptations, and neuro-mechanical capacities. This imbalance progressively lowers the pain trigger threshold and explains clinical variability and strong dependence on posture and exposure duration (Labat et al., 2008).
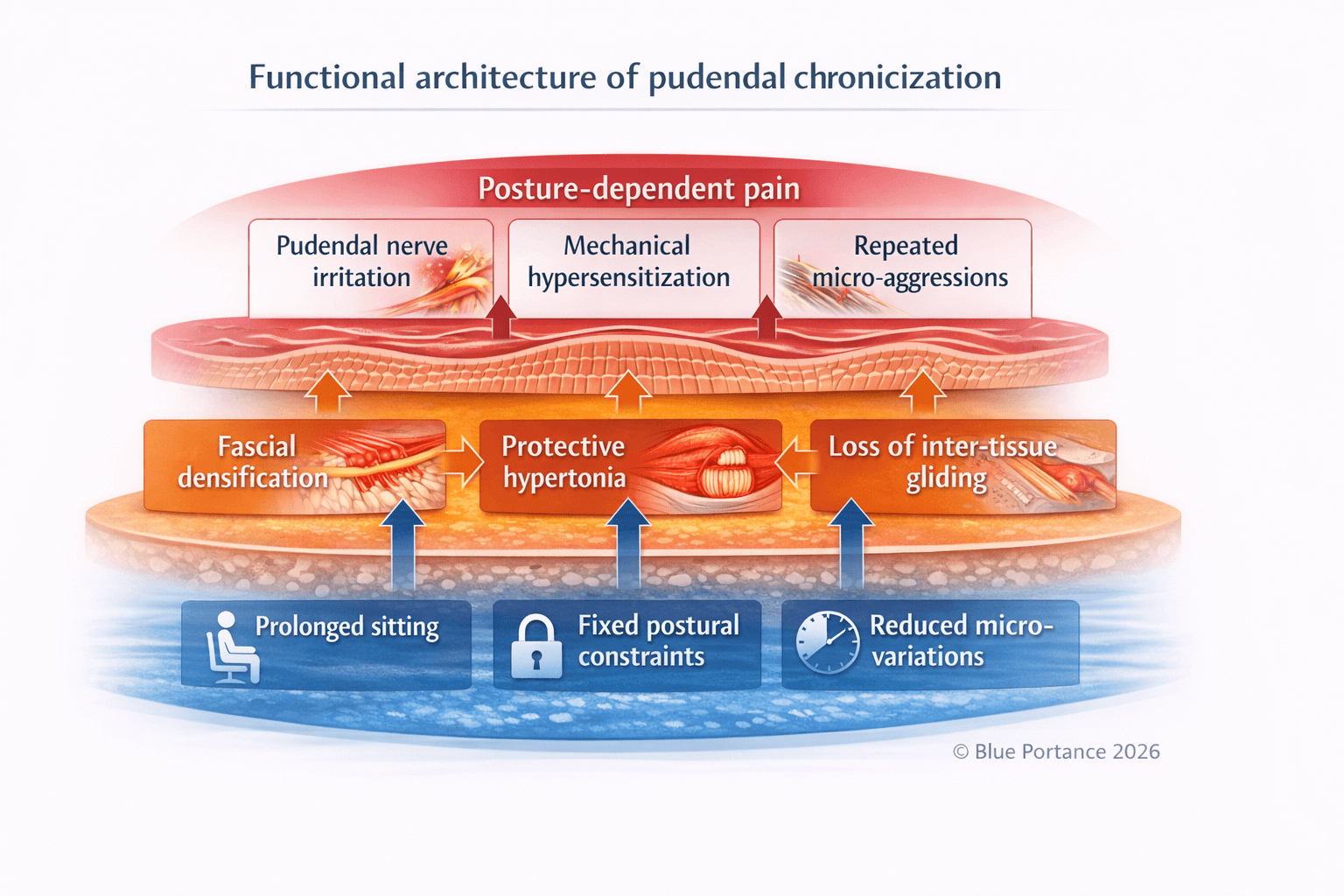
This model explains why pudendal neuralgia frequently escapes purely structural approaches and why imaging may remain normal despite severe symptoms. As long as the global mechanical regime remains unchanged, tissue and neuromuscular adaptations continue to maintain nerve irritation, independently of any localized intervention (Stecco & Stecco, 2024).
Chronicization thus appears not as an unexplained fatality, but as the coherent expression of a system that has lost its capacity for mechanical adaptation. This functional reading constitutes an essential prerequisite for any strategy aimed at interrupting self-sustaining pain loops, by acting not on a single point, but on the mechanical regime as a whole.
Scientific references
- Stecco A., Stecco C. (2024). Fascial densification and neuropathic pain: clinical implications.
- Butler D. (2000). The Sensitive Nervous System. Noigroup Publications.
- Cholewa J. et al. (2024). Myofascial entrapment in Alcock’s canal.
- Robert R. et al. (1998). Anatomical basis of pudendal nerve entrapment.
- Blue Portance — SBNFA™ Model (2026). NEURO-ANATOMY — Part V.


